Answer Summary:
The 2026 Competitive Bidding Program introduces formal “Remote Item Delivery” (RID) networks that cover large multi-state regions. To participate, DME providers must hold a valid “Non-Resident DME Permit” in every state within that region—not just their home state. Operating or bidding without these “foreign” state licenses is the #1 cause of immediate disqualification. This guide breaks down the “Strict State” requirements and the timeline for securing a 50-state footprint.
Introduction:
For years, local DME providers could ignore the licensing laws of neighboring states. If you didn’t have a physical office there, you simply shipped the box and didn’t worry about it.
2026 changes everything.
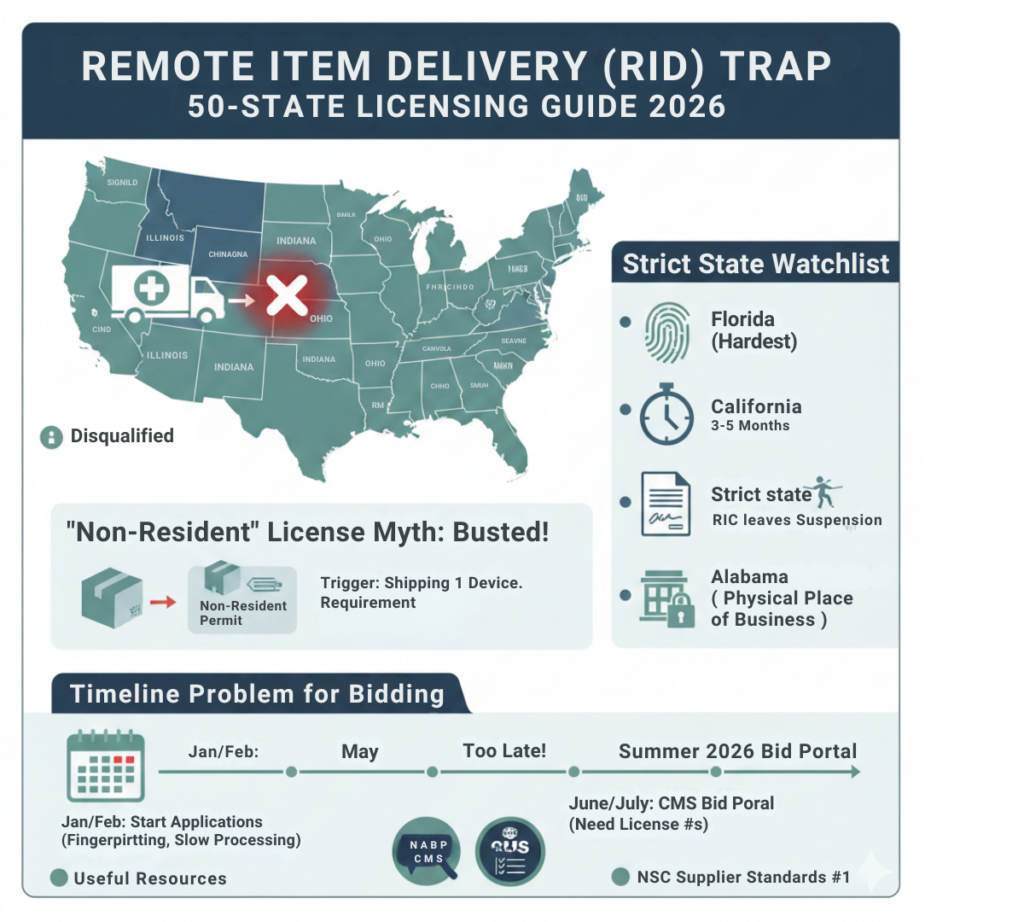
With the new Remote Item Delivery (RID) categories, CMS is asking a simple question: “Can you legally ship to every patient in this region?”
If you are bidding for the “Midwest Region” (which might include Illinois, Indiana, Ohio, and Michigan), and you only hold an Illinois license, you are disqualified. You cannot “cherry-pick” states within a defined CMS region.
Furthermore, state pharmacy boards have become aggressive. They are issuing cease-and-desist orders to out-of-state DMEs that ship direct-to-patient without a local permit.
The “Non-Resident” License Myth
Many providers believe: “I don’t have a building in Ohio, so I don’t need an Ohio license.”
False.
Most State Boards of Pharmacy (which regulate DME) have a specific license type called the “Non-Resident DME Permit” (or “Out-of-State Wholesaler/Retailer”).
- The Trigger: Shipping one prescriptive device into the state.
- The Requirement: You must maintain a valid license in your home state, and then apply for reciprocity in the targetstate.
The “Strict State” Watchlist
Not all states are created equal. While some just require a $200 fee, others require rigorous vetting.
1. Florida (The Hardest)
- Agency: AHCA (Agency for Health Care Administration).
- Requirement: Fingerprinting for all owners and the administrator. Extensive background checks.
- Timeline: 3–5 Months.
2. California (The Strictest)
- Agency: Board of Pharmacy.
- Requirement: You must designate a “Representative-in-Charge” (RIC).
- Trap: If your RIC leaves the company, you must replace them immediately or your license is suspended.
3. Alabama & Tennessee
- Requirement: Often require a specific “Physical Place of Business” affidavit, even for mail-order companies.
The Timeline Problem for Bidding
If you intend to bid in Summer 2026, you cannot wait until May to apply for these licenses.
- Fingerprinting: Requires mailing physical cards or visiting specific vendor locations.
- Processing: State Boards are notoriously slow.
- The Reality: If you don’t start the application process in January/February, you will not have the license numbers in time to enter them into the CMS Bid Portal.
Useful Resources for Licensing
- NABP (National Association of Boards of Pharmacy): Search for the specific “DME” or “Medical Device” rules in your target states. National Association of Boards of Pharmacy (NABP) Search
- NSC Supplier Standards: Review Standard #1, which mandates compliance with all state licensure laws CMS NSC Standards
WWS Value Proposition:
We Are Your Licensing Architects
Building a multi-state footprint is complex. Wonder Worth Solutions manages the entire Multi-State Licensure Project.
- Gap Analysis: We tell you exactly which licenses you need for your target regions.
- Application Management: We handle the fingerprinting logistics, notary requirements, and 50-state submission packets.
- Renewals: We track the expiration dates so you never drop a state.
Planning to bid regionally?
Let’s check your map. for a Multi-State License Feasibility Study to ensure you are legal in every zip code you serve.




